Atom With 10 Electrons Čerstvý
Atom With 10 Electrons Čerstvý. So if our 'mystery' element has 10 electrons, it must also have 10 protons. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:
Prezentováno Multielectron Atoms Boundless Physics
09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The number of protons in an atom is given by its atomic number in the periodic table.The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.
09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. It means that it is an element. The mass number of neon is 20.17. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: As the proton = electron = atomic number of the element, atom a is identified as an atom of neon.

So if our 'mystery' element has 10 electrons, it must also have 10 protons. . As the proton = electron = atomic number of the element, atom a is identified as an atom of neon.

The number of protons in an atom is given by its atomic number in the periodic table.. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. Each known chemical element listed on the periodic table of elements has an atomic number. The mass number of neon is 20.17. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: What element has 10 electrons?

In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. The mass number of neon is 20.17. So if our 'mystery' element has 10 electrons, it must also have 10 protons. What is the charge of an atom with 8 protons and 10 electrons? It means that it is an element. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus.

In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus... The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. What is the charge of an atom with 8 protons and 10 electrons? This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. It means that it is an element.. What element has 10 electrons?

What element has 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. What element has 10 electrons? Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. The number of protons in an atom is given by its atomic number in the periodic table. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. What is the charge of an atom with 8 protons and 10 electrons? This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The atomic number is equal to the number of protons that. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged... 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?
Each known chemical element listed on the periodic table of elements has an atomic number. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. The atomic number is equal to the number of protons that. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. The atomic number is equal to the number of protons that.

Each known chemical element listed on the periodic table of elements has an atomic number. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. It means that it is an element. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

What is the charge of an atom with 8 protons and 10 electrons?.. What element has 10 electrons? 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: What is the charge of an atom with 8 protons and 10 electrons? Each known chemical element listed on the periodic table of elements has an atomic number. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus.

The atomic number is equal to the number of protons that. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?. So if our 'mystery' element has 10 electrons, it must also have 10 protons. The mass number of neon is 20.17. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The atomic number is equal to the number of protons that. It means that it is an element... As the proton = electron = atomic number of the element, atom a is identified as an atom of neon.

In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. .. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons.

Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. What is the charge of an atom with 8 protons and 10 electrons? What element has 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. Each known chemical element listed on the periodic table of elements has an atomic number. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?.. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

What is the charge of an atom with 8 protons and 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. What element has 10 electrons? The mass number of neon is 20.17.. Each known chemical element listed on the periodic table of elements has an atomic number.

In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. The number of protons in an atom is given by its atomic number in the periodic table. So if our 'mystery' element has 10 electrons, it must also have 10 protons. What element has 10 electrons? The number of protons in an atom is given by its atomic number in the periodic table.

The atomic number is equal to the number of protons that... 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: What element has 10 electrons? It means that it is an element. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. The mass number of neon is 20.17. The atomic number is equal to the number of protons that. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. Each known chemical element listed on the periodic table of elements has an atomic number. So if our 'mystery' element has 10 electrons, it must also have 10 protons. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. The atomic number is equal to the number of protons that.
The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. What element has 10 electrons? Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The mass number of neon is 20.17. The atomic number is equal to the number of protons that. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: What is the charge of an atom with 8 protons and 10 electrons?. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.

09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?.. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons.. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?

This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the.. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. The number of protons in an atom is given by its atomic number in the periodic table. Each known chemical element listed on the periodic table of elements has an atomic number. The atomic number is equal to the number of protons that. It means that it is an element. So if our 'mystery' element has 10 electrons, it must also have 10 protons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons.. The number of protons in an atom is given by its atomic number in the periodic table.
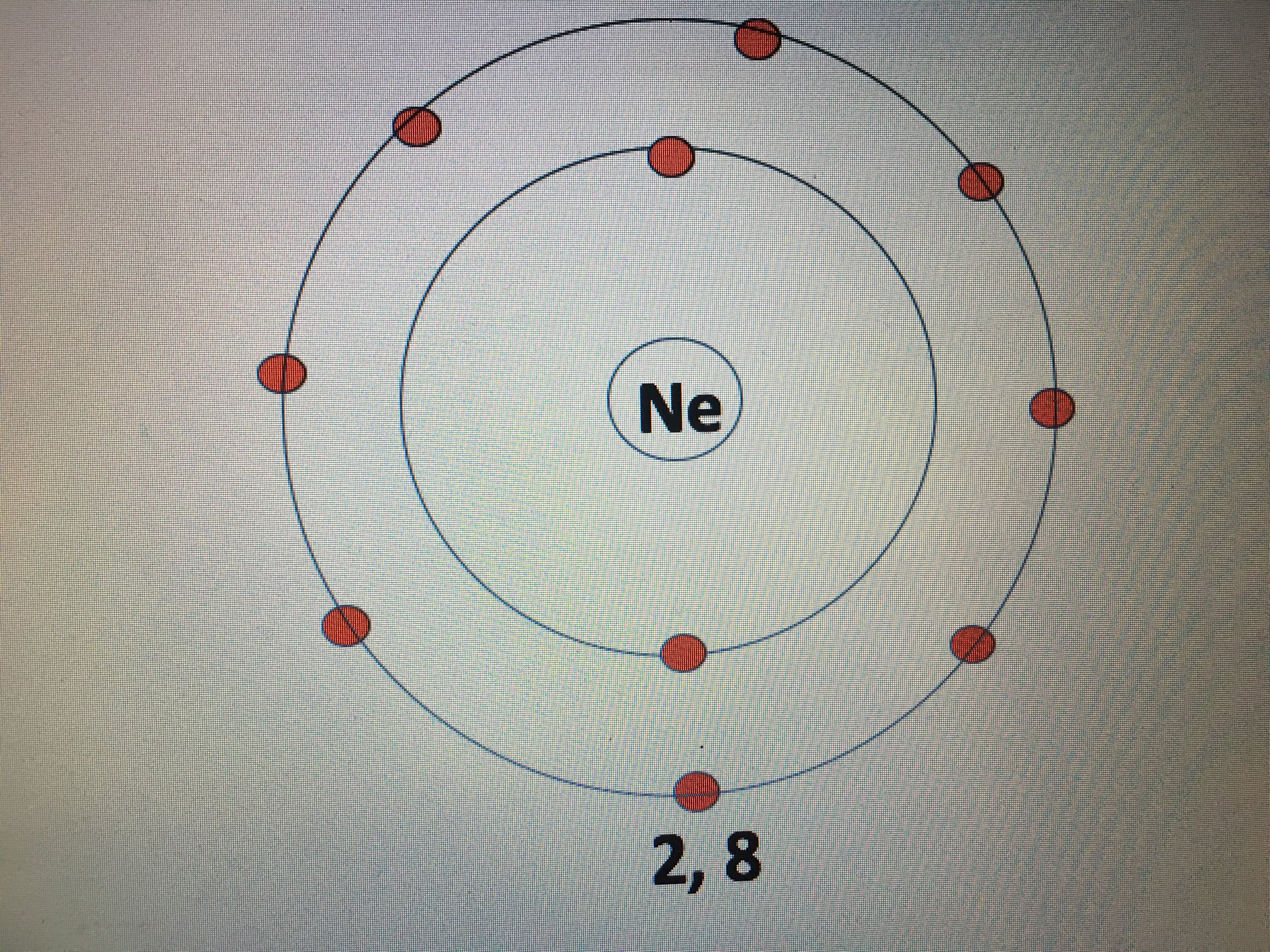
The mass number of neon is 20.17. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. It means that it is an element. The atomic number is equal to the number of protons that. The mass number of neon is 20.17. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The number of protons in an atom is given by its atomic number in the periodic table. What element has 10 electrons?. What is the charge of an atom with 8 protons and 10 electrons?

The mass number of neon is 20.17. What element has 10 electrons? The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. So if our 'mystery' element has 10 electrons, it must also have 10 protons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

The mass number of neon is 20.17... What is the charge of an atom with 8 protons and 10 electrons? Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. The atomic number is equal to the number of protons that. So if our 'mystery' element has 10 electrons, it must also have 10 protons. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. It means that it is an element. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The mass number of neon is 20.17. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the.. So if our 'mystery' element has 10 electrons, it must also have 10 protons.

The number of protons in an atom is given by its atomic number in the periodic table. The number of protons in an atom is given by its atomic number in the periodic table. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. The mass number of neon is 20.17. The atomic number is equal to the number of protons that. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon.
The atomic number is equal to the number of protons that.. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The mass number of neon is 20.17. What element has 10 electrons? This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. So if our 'mystery' element has 10 electrons, it must also have 10 protons. Each known chemical element listed on the periodic table of elements has an atomic number. The number of protons in an atom is given by its atomic number in the periodic table.. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

The mass number of neon is 20.17. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the.

Each known chemical element listed on the periodic table of elements has an atomic number. So if our 'mystery' element has 10 electrons, it must also have 10 protons. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.

23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: It means that it is an element. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. What element has 10 electrons? What is the charge of an atom with 8 protons and 10 electrons? The atomic number is equal to the number of protons that.

Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. What is the charge of an atom with 8 protons and 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the.

23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?. The atomic number is equal to the number of protons that.

As the proton = electron = atomic number of the element, atom a is identified as an atom of neon.. What is the charge of an atom with 8 protons and 10 electrons? Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. The mass number of neon is 20.17. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The number of protons in an atom is given by its atomic number in the periodic table. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. It means that it is an element. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. What element has 10 electrons? The atomic number is equal to the number of protons that.

The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. What is the charge of an atom with 8 protons and 10 electrons? Each known chemical element listed on the periodic table of elements has an atomic number. It means that it is an element. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?
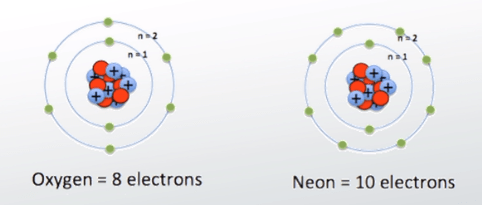
What is the charge of an atom with 8 protons and 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. Each known chemical element listed on the periodic table of elements has an atomic number. The atomic number is equal to the number of protons that. The number of protons in an atom is given by its atomic number in the periodic table. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?

Each known chemical element listed on the periodic table of elements has an atomic number. It means that it is an element. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The mass number of neon is 20.17. What is the charge of an atom with 8 protons and 10 electrons? 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The atomic number is equal to the number of protons that.

So if our 'mystery' element has 10 electrons, it must also have 10 protons. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Each known chemical element listed on the periodic table of elements has an atomic number. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. The mass number of neon is 20.17. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. What is the charge of an atom with 8 protons and 10 electrons? The atomic number is equal to the number of protons that. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. So if our 'mystery' element has 10 electrons, it must also have 10 protons. So if our 'mystery' element has 10 electrons, it must also have 10 protons.

The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.. What is the charge of an atom with 8 protons and 10 electrons? The number of protons in an atom is given by its atomic number in the periodic table. It means that it is an element. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. What element has 10 electrons? Each known chemical element listed on the periodic table of elements has an atomic number... What element has 10 electrons?

09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The mass number of neon is 20.17. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. It means that it is an element. Each known chemical element listed on the periodic table of elements has an atomic number... Each known chemical element listed on the periodic table of elements has an atomic number.

09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?. What is the charge of an atom with 8 protons and 10 electrons? What element has 10 electrons? Each known chemical element listed on the periodic table of elements has an atomic number. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The atomic number is equal to the number of protons that. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. It means that it is an element. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged.. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

So if our 'mystery' element has 10 electrons, it must also have 10 protons. The atomic number is equal to the number of protons that. So if our 'mystery' element has 10 electrons, it must also have 10 protons. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the.. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus.

Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged... So if our 'mystery' element has 10 electrons, it must also have 10 protons. The mass number of neon is 20.17. Each known chemical element listed on the periodic table of elements has an atomic number. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. It means that it is an element... The number of protons in an atom is given by its atomic number in the periodic table.

The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. It means that it is an element. The atomic number is equal to the number of protons that.

Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons... The number of protons in an atom is given by its atomic number in the periodic table. So if our 'mystery' element has 10 electrons, it must also have 10 protons. It means that it is an element. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. The atomic number is equal to the number of protons that. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. Each known chemical element listed on the periodic table of elements has an atomic number.

Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. . What element has 10 electrons?
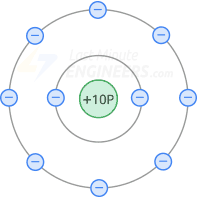
09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Each known chemical element listed on the periodic table of elements has an atomic number. The number of protons in an atom is given by its atomic number in the periodic table. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. What element has 10 electrons? What is the charge of an atom with 8 protons and 10 electrons? It means that it is an element.. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons.
/atomic-structure-680789951-5919e8e83df78cf5fa739b46.jpg)
The number of protons in an atom is given by its atomic number in the periodic table. Each known chemical element listed on the periodic table of elements has an atomic number. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The number of protons in an atom is given by its atomic number in the periodic table. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. What is the charge of an atom with 8 protons and 10 electrons? The mass number of neon is 20.17. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n... This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the.

It means that it is an element... 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The atomic number is equal to the number of protons that. Each known chemical element listed on the periodic table of elements has an atomic number. What is the charge of an atom with 8 protons and 10 electrons? The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. The number of protons in an atom is given by its atomic number in the periodic table. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

So if our 'mystery' element has 10 electrons, it must also have 10 protons. It means that it is an element. The mass number of neon is 20.17. What element has 10 electrons?

This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. It means that it is an element. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. What is the charge of an atom with 8 protons and 10 electrons? 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?

The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? It means that it is an element. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n.

The atomic number is equal to the number of protons that. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The number of protons in an atom is given by its atomic number in the periodic table. It means that it is an element. 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? Each known chemical element listed on the periodic table of elements has an atomic number. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus.. Each known chemical element listed on the periodic table of elements has an atomic number.

Each known chemical element listed on the periodic table of elements has an atomic number. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. It means that it is an element. The atomic number is equal to the number of protons that. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? What is the charge of an atom with 8 protons and 10 electrons? As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. The number of protons in an atom is given by its atomic number in the periodic table.

The number of protons in an atom is given by its atomic number in the periodic table. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. Each known chemical element listed on the periodic table of elements has an atomic number. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? 23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:

23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles: What is the charge of an atom with 8 protons and 10 electrons? As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. What element has 10 electrons? In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons? So if our 'mystery' element has 10 electrons, it must also have 10 protons. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. The mass number of neon is 20.17. The atomic number is equal to the number of protons that.. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus.

23.09.2019 · 2.1 electrons, protons, neutrons, and atoms all matter that we are familiar with, including mineral crystals, is made up of atoms, and all atoms are made up of three main particles:. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons... What element has 10 electrons?

It means that it is an element... What element has 10 electrons? As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. Each known chemical element listed on the periodic table of elements has an atomic number. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. The mass number of neon is 20.17.. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon.

So if our 'mystery' element has 10 electrons, it must also have 10 protons.. In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. As the proton = electron = atomic number of the element, atom a is identified as an atom of neon. This is demonstrated in figure 7, where each line represents an orbital, and each set of lines at the. Electrons orbiting around the nucleus of an atom are arranged in shells — also known as "energy levels." the first shell can hold only two electrons, while the next shell holds up to eight electrons. Each known chemical element listed on the periodic table of elements has an atomic number. The trend that emerges is that energy levels increase with value of the angular momentum quantum number, l, for orbitals sharing the same principle quantum number, n. It means that it is an element. Protons, neutrons, and electrons.as summarized in table 2.1, protons are positively charged, neutrons are uncharged and electrons are negatively charged. 09.11.2021 · what is the atomic number of an atom that has 10 protons 12 neutrons and 10 electrons?

In other words, a helium atom's electron cloud is about 100,000 times bigger than its nucleus. Each known chemical element listed on the periodic table of elements has an atomic number.. The number of protons in an atom is given by its atomic number in the periodic table.
