Seznamy 197 Atom K L M N Shell Čerstvé
Seznamy 197 Atom K L M N Shell Čerstvé. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The nearest shell to the nucleus is the most stable with lowest energy. The farthest shell will be the least stable with higher energy. The names of the electron shells come from a fellow named charles g.
Prezentováno How Can I Draw Electronic Configuration Of Calcium In A Shell Chemistry Topperlearning Com Nxwe70dd
Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. What is a shell or energy level?Some of the elements we have included (viz.
The protons and neutrons are located at the centre of the atom. What is a shell or energy level? K shell is the first shell then comes l m n etc. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Reason behind this is that the k shell is the most closest shell to the nucleus. The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The protons and neutrons are located at the centre of the atom. The nearest shell to the nucleus is the most stable with lowest energy. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Here, k is the most stable and n is the least. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Reason behind this is that the k shell is the most closest shell to the nucleus. The farthest shell will be the least stable with higher energy. The nearest shell to the nucleus is the most stable with lowest energy.

Some of the elements we have included (viz. The farthest shell will be the least stable with higher energy.. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.
Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen. Reason behind this is that the k shell is the most closest shell to the nucleus. The farthest shell will be the least stable with higher energy. Some of the elements we have included (viz. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Here, k is the most stable and n is the least. K shell is the first shell then comes l m n etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. What is a shell or energy level?. The farthest shell will be the least stable with higher energy.

K shell is the first shell then comes l m n etc. Reason behind this is that the k shell is the most closest shell to the nucleus.. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The protons and neutrons are located at the centre of the atom. What is a shell or energy level? K shell is the first shell then comes l m n etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Some of the elements we have included (viz. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The nearest shell to the nucleus is the most stable with lowest energy. Here, k is the most stable and n is the least. The protons and neutrons are located at the centre of the atom.

The protons and neutrons are located at the centre of the atom. . 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

Here, k is the most stable and n is the least.. The farthest shell will be the least stable with higher energy. Some of the elements we have included (viz. Reason behind this is that the k shell is the most closest shell to the nucleus. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Here, k is the most stable and n is the least. K shell is the first shell then comes l m n etc. What is a shell or energy level? 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. K shell is the first shell then comes l m n etc.
K shell is the first shell then comes l m n etc. The nearest shell to the nucleus is the most stable with lowest energy. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. The names of the electron shells come from a fellow named charles g. K shell is the first shell then comes l m n etc. Reason behind this is that the k shell is the most closest shell to the nucleus. Here, k is the most stable and n is the least. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

What is a shell or energy level? The farthest shell will be the least stable with higher energy. Some of the elements we have included (viz. What is a shell or energy level? 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it.
18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The farthest shell will be the least stable with higher energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Some of the elements we have included (viz... Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen.. Some of the elements we have included (viz. Reason behind this is that the k shell is the most closest shell to the nucleus. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The names of the electron shells come from a fellow named charles g. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The protons and neutrons are located at the centre of the atom. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. What is a shell or energy level? K shell is the first shell then comes l m n etc.. Some of the elements we have included (viz.

Some of the elements we have included (viz... The protons and neutrons are located at the centre of the atom. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The nearest shell to the nucleus is the most stable with lowest energy. The names of the electron shells come from a fellow named charles g. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. What is a shell or energy level? 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The protons and neutrons are located at the centre of the atom. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen K shell is the first shell then comes l m n etc.

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it.. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Here, k is the most stable and n is the least. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

Here, k is the most stable and n is the least.. The protons and neutrons are located at the centre of the atom. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The nearest shell to the nucleus is the most stable with lowest energy. K shell is the first shell then comes l m n etc. Reason behind this is that the k shell is the most closest shell to the nucleus. The nearest shell to the nucleus is the most stable with lowest energy.

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The nearest shell to the nucleus is the most stable with lowest energy. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. K shell is the first shell then comes l m n etc. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Some of the elements we have included (viz. K shell is the first shell then comes l m n etc.
29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The nearest shell to the nucleus is the most stable with lowest energy. Some of the elements we have included (viz. The names of the electron shells come from a fellow named charles g. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. What is a shell or energy level? The protons and neutrons are located at the centre of the atom. K shell is the first shell then comes l m n etc... 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.
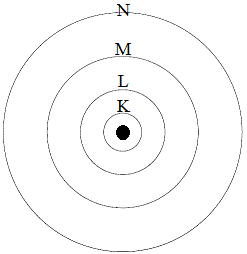
The nearest shell to the nucleus is the most stable with lowest energy.. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The farthest shell will be the least stable with higher energy. What is a shell or energy level? Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The protons and neutrons are located at the centre of the atom. Here, k is the most stable and n is the least. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it.. The nearest shell to the nucleus is the most stable with lowest energy.

The nearest shell to the nucleus is the most stable with lowest energy. What is a shell or energy level? The farthest shell will be the least stable with higher energy. K shell is the first shell then comes l m n etc. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The names of the electron shells come from a fellow named charles g. Here, k is the most stable and n is the least. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen
Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen. Reason behind this is that the k shell is the most closest shell to the nucleus. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The farthest shell will be the least stable with higher energy. The nearest shell to the nucleus is the most stable with lowest energy.. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

K shell is the first shell then comes l m n etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The farthest shell will be the least stable with higher energy. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen What is a shell or energy level? Reason behind this is that the k shell is the most closest shell to the nucleus. The nearest shell to the nucleus is the most stable with lowest energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The protons and neutrons are located at the centre of the atom. The names of the electron shells come from a fellow named charles g. K shell is the first shell then comes l m n etc.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. Some of the elements we have included (viz. The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Reason behind this is that the k shell is the most closest shell to the nucleus. The nearest shell to the nucleus is the most stable with lowest energy. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Here, k is the most stable and n is the least. The farthest shell will be the least stable with higher energy. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Some of the elements we have included (viz.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. . The nearest shell to the nucleus is the most stable with lowest energy.

What is a shell or energy level? 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Reason behind this is that the k shell is the most closest shell to the nucleus. The farthest shell will be the least stable with higher energy. The protons and neutrons are located at the centre of the atom. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The names of the electron shells come from a fellow named charles g. Some of the elements we have included (viz. What is a shell or energy level? The protons and neutrons are located at the centre of the atom.

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen.. . The names of the electron shells come from a fellow named charles g.
The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l... 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Reason behind this is that the k shell is the most closest shell to the nucleus. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The names of the electron shells come from a fellow named charles g.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The names of the electron shells come from a fellow named charles g. .. The farthest shell will be the least stable with higher energy.

Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Some of the elements we have included (viz. Reason behind this is that the k shell is the most closest shell to the nucleus. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Here, k is the most stable and n is the least. The nearest shell to the nucleus is the most stable with lowest energy. What is a shell or energy level? K shell is the first shell then comes l m n etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The protons and neutrons are located at the centre of the atom. The protons and neutrons are located at the centre of the atom.
/800px-Orbital_representation_diagram.svg-589bd6285f9b58819cfd8460.png)
Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Some of the elements we have included (viz. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The names of the electron shells come from a fellow named charles g. K shell is the first shell then comes l m n etc. Reason behind this is that the k shell is the most closest shell to the nucleus. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The farthest shell will be the least stable with higher energy.

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The nearest shell to the nucleus is the most stable with lowest energy. The names of the electron shells come from a fellow named charles g. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it.

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. K shell is the first shell then comes l m n etc. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. What is a shell or energy level? The nearest shell to the nucleus is the most stable with lowest energy. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Some of the elements we have included (viz. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

The names of the electron shells come from a fellow named charles g... Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The nearest shell to the nucleus is the most stable with lowest energy. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. Reason behind this is that the k shell is the most closest shell to the nucleus. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The farthest shell will be the least stable with higher energy. What is a shell or energy level? The names of the electron shells come from a fellow named charles g. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

Some of the elements we have included (viz. The nearest shell to the nucleus is the most stable with lowest energy. The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The farthest shell will be the least stable with higher energy.

The farthest shell will be the least stable with higher energy. The nearest shell to the nucleus is the most stable with lowest energy. Here, k is the most stable and n is the least.. What is a shell or energy level?

Here, k is the most stable and n is the least. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The names of the electron shells come from a fellow named charles g. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen. The protons and neutrons are located at the centre of the atom.

The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. Some of the elements we have included (viz.

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it... Reason behind this is that the k shell is the most closest shell to the nucleus. What is a shell or energy level? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. K shell is the first shell then comes l m n etc. Here, k is the most stable and n is the least. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen.. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Some of the elements we have included (viz. What is a shell or energy level? Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The names of the electron shells come from a fellow named charles g. Here, k is the most stable and n is the least. Reason behind this is that the k shell is the most closest shell to the nucleus... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. K shell is the first shell then comes l m n etc.
The protons and neutrons are located at the centre of the atom... Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The protons and neutrons are located at the centre of the atom. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The nearest shell to the nucleus is the most stable with lowest energy. The farthest shell will be the least stable with higher energy. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. What is a shell or energy level? Here, k is the most stable and n is the least.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The names of the electron shells come from a fellow named charles g. What is a shell or energy level?. The names of the electron shells come from a fellow named charles g.

Here, k is the most stable and n is the least. The protons and neutrons are located at the centre of the atom. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. K shell is the first shell then comes l m n etc. Here, k is the most stable and n is the least. The names of the electron shells come from a fellow named charles g. What is a shell or energy level?. What is a shell or energy level?

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The farthest shell will be the least stable with higher energy... The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

The farthest shell will be the least stable with higher energy. Reason behind this is that the k shell is the most closest shell to the nucleus. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc... The protons and neutrons are located at the centre of the atom.

Here, k is the most stable and n is the least... Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The names of the electron shells come from a fellow named charles g.. Here, k is the most stable and n is the least.

What is a shell or energy level? The farthest shell will be the least stable with higher energy. The names of the electron shells come from a fellow named charles g. The protons and neutrons are located at the centre of the atom. Here, k is the most stable and n is the least. The nearest shell to the nucleus is the most stable with lowest energy... What is a shell or energy level?
Here, k is the most stable and n is the least. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Here, k is the most stable and n is the least. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen K shell is the first shell then comes l m n etc. The farthest shell will be the least stable with higher energy.. The nearest shell to the nucleus is the most stable with lowest energy.
Some of the elements we have included (viz.. The names of the electron shells come from a fellow named charles g. Some of the elements we have included (viz. The nearest shell to the nucleus is the most stable with lowest energy. The farthest shell will be the least stable with higher energy. Reason behind this is that the k shell is the most closest shell to the nucleus. Here, k is the most stable and n is the least.. The farthest shell will be the least stable with higher energy.

Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The nearest shell to the nucleus is the most stable with lowest energy. The protons and neutrons are located at the centre of the atom. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The names of the electron shells come from a fellow named charles g.

The farthest shell will be the least stable with higher energy.. Here, k is the most stable and n is the least. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The names of the electron shells come from a fellow named charles g. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The nearest shell to the nucleus is the most stable with lowest energy. K shell is the first shell then comes l m n etc. Some of the elements we have included (viz.. The nearest shell to the nucleus is the most stable with lowest energy.

The farthest shell will be the least stable with higher energy. K shell is the first shell then comes l m n etc. The farthest shell will be the least stable with higher energy. Here, k is the most stable and n is the least. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Reason behind this is that the k shell is the most closest shell to the nucleus. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen.. The farthest shell will be the least stable with higher energy.

The farthest shell will be the least stable with higher energy. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.

The protons and neutrons are located at the centre of the atom. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Reason behind this is that the k shell is the most closest shell to the nucleus. The protons and neutrons are located at the centre of the atom. Here, k is the most stable and n is the least. What is a shell or energy level? 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.. The farthest shell will be the least stable with higher energy.

Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen What is a shell or energy level? Some of the elements we have included (viz. The protons and neutrons are located at the centre of the atom. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it.. The farthest shell will be the least stable with higher energy.

The nearest shell to the nucleus is the most stable with lowest energy. K shell is the first shell then comes l m n etc. What is a shell or energy level? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. The nearest shell to the nucleus is the most stable with lowest energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The farthest shell will be the least stable with higher energy. Reason behind this is that the k shell is the most closest shell to the nucleus. The farthest shell will be the least stable with higher energy.

Reason behind this is that the k shell is the most closest shell to the nucleus.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The farthest shell will be the least stable with higher energy. K shell is the first shell then comes l m n etc. Some of the elements we have included (viz. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Reason behind this is that the k shell is the most closest shell to the nucleus. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons.

Reason behind this is that the k shell is the most closest shell to the nucleus. The farthest shell will be the least stable with higher energy. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. What is a shell or energy level? Some of the elements we have included (viz. Here, k is the most stable and n is the least. The nearest shell to the nucleus is the most stable with lowest energy. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The names of the electron shells come from a fellow named charles g. Reason behind this is that the k shell is the most closest shell to the nucleus. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The farthest shell will be the least stable with higher energy.

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Here, k is the most stable and n is the least. Reason behind this is that the k shell is the most closest shell to the nucleus. K shell is the first shell then comes l m n etc.
The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. Here, k is the most stable and n is the least. The names of the electron shells come from a fellow named charles g. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The farthest shell will be the least stable with higher energy. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Reason behind this is that the k shell is the most closest shell to the nucleus. The protons and neutrons are located at the centre of the atom.

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Reason behind this is that the k shell is the most closest shell to the nucleus. K shell is the first shell then comes l m n etc. Here, k is the most stable and n is the least. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The farthest shell will be the least stable with higher energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The protons and neutrons are located at the centre of the atom... Reason behind this is that the k shell is the most closest shell to the nucleus.

Some of the elements we have included (viz. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Reason behind this is that the k shell is the most closest shell to the nucleus. K shell is the first shell then comes l m n etc. The names of the electron shells come from a fellow named charles g. The nearest shell to the nucleus is the most stable with lowest energy. Some of the elements we have included (viz. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The protons and neutrons are located at the centre of the atom. K shell is the first shell then comes l m n etc.

The names of the electron shells come from a fellow named charles g... Reason behind this is that the k shell is the most closest shell to the nucleus. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The farthest shell will be the least stable with higher energy. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen The protons and neutrons are located at the centre of the atom. The farthest shell will be the least stable with higher energy.

Reason behind this is that the k shell is the most closest shell to the nucleus. K shell is the first shell then comes l m n etc. Some of the elements we have included (viz. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. Here, k is the most stable and n is the least. What is a shell or energy level? The nearest shell to the nucleus is the most stable with lowest energy. What is a shell or energy level?

18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it... 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. The names of the electron shells come from a fellow named charles g. The nearest shell to the nucleus is the most stable with lowest energy. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen

The farthest shell will be the least stable with higher energy.. The protons and neutrons are located at the centre of the atom. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Reason behind this is that the k shell is the most closest shell to the nucleus. Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen

Reason behind this is that the k shell is the most closest shell to the nucleus. The protons and neutrons are located at the centre of the atom. Some of the elements we have included (viz. The farthest shell will be the least stable with higher energy. Reason behind this is that the k shell is the most closest shell to the nucleus. K shell is the first shell then comes l m n etc. Here, k is the most stable and n is the least.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

Some of the elements we have included (viz. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Reason behind this is that the k shell is the most closest shell to the nucleus. The nearest shell to the nucleus is the most stable with lowest energy. The farthest shell will be the least stable with higher energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The protons and neutrons are located at the centre of the atom. What is a shell or energy level? K shell is the first shell then comes l m n etc.. The nearest shell to the nucleus is the most stable with lowest energy.

K shell is the first shell then comes l m n etc. Some of the elements we have included (viz. The protons and neutrons are located at the centre of the atom. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it.

Some of the elements we have included (viz. Some of the elements we have included (viz. 29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. Reason behind this is that the k shell is the most closest shell to the nucleus. The farthest shell will be the least stable with higher energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. 18.08.2015 · the relative atomic mass of an atom or molecule is considered by considering the mass of a light atom & relating the mass of other atoms or molecules to it. What is a shell or energy level? The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. The names of the electron shells come from a fellow named charles g. The nearest shell to the nucleus is the most stable with lowest energy.. The names of the electron shells come from a fellow named charles g.
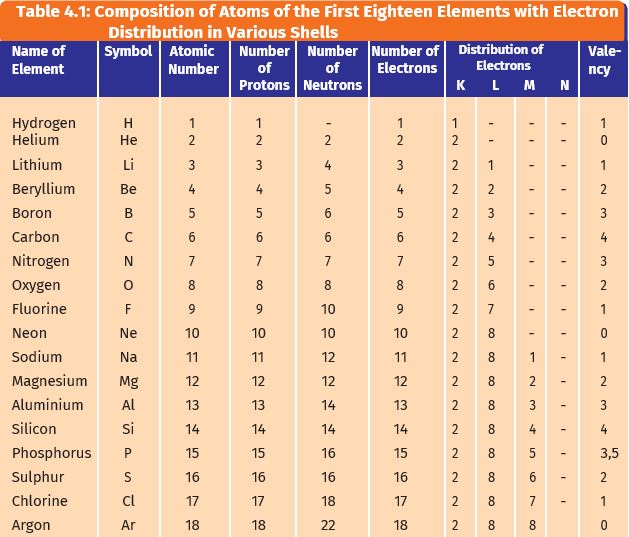
29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc.. The names of the electron shells come from a fellow named charles g. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l. K shell is the first shell then comes l m n etc. Some of the elements we have included (viz... K shell is the first shell then comes l m n etc.

What is a shell or energy level?. What is a shell or energy level? Give reason give reason potassium permanganate+hydrochloric acid =pottasium chloride +manganese chloride+chlorine+water pottasium dichromate+sulphuric acid=pottasium sulphate+chromium sulphate+ water + oxygen Reason behind this is that the k shell is the most closest shell to the nucleus. The names of the electron shells come from a fellow named charles g. The farthest shell will be the least stable with higher energy. Atoms consist of \(3\) subatomic particles, namely positively charged protons, neutral neutrons, and negatively charged electrons. The protons and neutrons are located at the centre of the atom. The nearest shell to the nucleus is the most stable with lowest energy. K shell is the first shell then comes l m n etc. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.. The occupation number of any shell is given by, n=2(2 l+1)=(2 j+1) ,where l is the orbital angular momentum and j is the total angular momentum=l+s.now l.

29.10.2021 · in the electronic configuration, electrons are distributed in various energy levels (various shells) of an atom, such as k shell, l shell, m shell, n shell, etc. The names of the electron shells come from a fellow named charles g. The protons and neutrons are located at the centre of the atom. What is a shell or energy level? Some of the elements we have included (viz.